- Guideline article
- Open access
- Published:
EANM guideline for harmonisation on molar activity or specific activity of radiopharmaceuticals: impact on safety and imaging quality
EJNMMI Radiopharmacy and Chemistry volume 6, Article number: 34 (2021)
Abstract
This guideline on molar activity (Am) and specific activity (As) focusses on small molecules, peptides and macromolecules radiolabelled for diagnostic and therapeutic applications. In this guideline we describe the definition of Am and As, and how these measurements must be standardised and harmonised. Selected examples highlighting the importance of Am and As in imaging studies of saturable binding sites will be given, and the necessity of using appropriate materials and equipment will be discussed. Furthermore, common Am pitfalls and remedies are described. Finally, some aspects of Am in relation the emergence of a new generation of highly sensitive PET scanners will be discussed.
Rationale
Harmonisation and standardisation of radiopharmaceutical terms and conventions are necessary to allow radiopharmaceutical research to be unambiguously understood and methodologies tested and transferred between laboratories. The aim of this guideline on molar activity is to provide a review of basic concepts, practical considerations, pitfalls and recommendations for obtaining high molar activity (Am) or specific activity (As).
In the field of radiopharmaceutical sciences, small molecules, peptides and macromolecules radiolabelled for diagnostic and therapeutic applications are defined as radiolabelled compounds where a radionuclide is attached by covalent or ionic bonds to a non-radioactive molecular scaffold (precursor). A radiotracer is a radiolabelled compound that is often administered to a complex environment (e.g. a tissue or living organism) without perturbing the system under study.
Most radionuclides used for the production of radiotracers are obtained via ‘no-carrier’ added (n.c.a.) nuclear reactions (definitions for carrier and n.c.a. can be found in Ref. Coenen et al. 2017). However, fluctuations in the amount of carrier contamination from external sources and starting materials during the radiosynthesis can dilute the radiolabelled compound with its non-labelled analogue and thus decrease the Am or As of the product. Fluctuations in Am or As for different radiotracer productions may influence the kinetics and tissue uptake of the radiotracers and therefore the quality of the scan data. Increased mass administration can, for example, lead to pharmacological or toxicological effects. Therefore, it is important to describe in detail how Am and As are defined and to provide appropriate guidelines on how to measure it. Moreover, the overall goal is to introduce a quality standard for Am and As to standardise and harmonise the measurement and applicability within the radiochemical and pharmaceutical scientific community. Finally, this guideline provides some working examples and recommendations.
Definition of molar activity (Am) and specific activity (As)
Am is defined as the amount of radioactivity per unit mole of the element or compound, is expressed in Bq/mol or GBq/µmol and is used where the molecular weight of the labelled material is known. As is expressed in Bq/g or GBq/µg and is normally used for macromolecules (proteins and antibodies) when the molecular weight of the labelled material is unknown.
Precursor residues or other impurities may be present in a final radiopharmaceutical preparation thus the term apparent molar or specific activity (apparent Am and apparent As) was introduced (Coenen et al. 2017). Apparent Am and apparent As take into account the amount of non-radiolabelled, radiolabelled impurities and remaining precursor which may interfere with the mass determination of the parent radiotracer.
The additional terms “effective Am and effective As” address other (unknown) materials present in a prepared batch, which have the chemically, biological or pharmacological potential to compete with the radiotracer binding to the intended target. An additional bioanalytical method has to be included to determine the efficacy of a formulated batch, such as receptor or enzyme-binding assays.
Measurement of molar activity and specific activity.
Molar activity (Am) is the ratio between the amount of radioactivity of a solution containing a radiotracer (ActivityA*) and the sum of the quantity of the radioactive compound (nA*) plus the quantity of the corresponding isotopically stable compound (nA). The unit of Am is typically Bq/mol or GBq/µmol.
Here, the \(Activity_{A*}\) is the measure of radioactivity present in an exact volume of a formulation measured in becquerel (Bq) using an activity meter (e.g. dose calibrator) (Eq. 1). \(n_{A}\) is the quantity of the isotopically stable compound whereas \(n_{{A^{*} }}\) is the quantity of the radioactive compound in the same volume. For the determination of the quantity of \(n_{A}\), a validated analytical method is required. A reliable and robust HPLC method for the determination of Am or As includes the validation of the following parameters, as described by the ICH guidelines for analytical methods: accuracy, precision (repeatability), specificity, detection limit, quantification limit, linearity (calibration curve) and range (Balaram et al. 2011). High-performance liquid chromatography (HPLC) or ultra-performance liquid chromatography (UPLC) and a suitable detection system such as UV–Vis spectrometry are generally used. Other analytical tools include, for example, gas chromatography/mass spectrometry (GC/MS) or liquid chromatography/mass spectrometry (LC/MS). See also the EANM guideline on validation of analytical methods for radiopharmaceuticals for more information. https://ejnmmipharmchem.springeropen.com/articles/10.1186/s41181-019-0086-z/.
A calibration curve has to be set up in an appropriate range in order to measure the required amount of compound and should cover concentrations down to the limit of detection (LOD). In principle, the area under the curve (AUC) of, for example, a UV–Vis absorbance (chromatographic peak) describes the sum of the amount of the isotopically stable compound and radiolabelled compound, as the radiolabelled and isotopically stable compound share the same absorption behaviour. However, the quantity of radiolabelled compound (\(n_{{A^{*} }}\)) changes over time following decay of the concerned radionuclide. Assuming that the quantity of the radioactive compound (\(n_{{A^{*} }}\)) is per se usually insignificant compared to the associated total moles of analogous stable compound, the sum \(n_{A} + n_{{A^{*} }}\) is simplified to be equal to \(n_{A}\) (Eq. 1).
The maximum theoretical Am for PET radionuclides 15O, 13N, 11C, 68Ga and 18F is directly linked (inversely proportional) to their half-life, see Fig. 1. Radionuclides with longer half-lives have lower theoretical Am compared to shorter lived radionuclides.
Specific activity (As) is usually expressed as Bq/g or GBq/µg and can be calculated with the same procedure as described for Am having the \(Activity_{A*}\) measured in an exact volume of a formulation and the compound measured in µg or g in the same volume. \(m_{A}\) is the quantity of the isotopically stable compound whereas \(m_{{A^{*} }}\) is the quantity of the radioactive compound. Assuming that the quantity of the radioactive compound (\(m_{{A^{*} }}\)) is per se usually insignificant compared to the associated total mass of analogous stable compound, the sum \(m_{A} + m_{{A^{*} }}\) is simplified to be equal to \(m_{A}\).
As can also be obtained by dividing Am by the molecular weight (MW—g/mol) of the compound, if this is known (Eq. 2).
Apparent molar activity
The apparent molar activity (apparent Am) takes into account the amounts of the radiolabelled (compound A*) and non-radiolabelled radiotracer (compound A), radiolabelled impurities (compound B*) and non-radiolabelled impurities (compound B) and remaining precursor (compound C) present expressed in mol (or µmol). Apparent Am is expressed as Bq/mol or GBq/µmol.
The amount of \(n_{A} ,\) \(n_{B}\) and \(n_{C}\) are determined by a quantitative analysis using calibration curves for A, B and C. The amount of unknown impurities (\(n_{B}\)) can be difficult to quantify and therefore calculated Am values may be somewhat uncertain. In practice, the unknown impurities are calculated using the absorption coefficient of the intended tracer.
For high quality radiotracer production, it is often mandatory to obtain radiochemical purities ≥ 95% and therefore radiochemical impurities should play a minor role in practice for the calculation of the apparent Am. However, non-radioactive scaffolds in routine production represent a separation problem, as for example precursors and peptides with chelators occupied with non-radioactive metal ions share similar retention times.
An different approach of Am calculation specific for radio-metals like gallium-68 labelled compounds is published by the IAEA “Gallium-68 cyclotron productions/TECDOC-1863 (annex 1 pp. 43–45)”. https://www.iaea.org/publications/13484/gallium-68-cyclotron-production
Description of a procedure to calculate the Am for a carbon-11 radiotracer using an HPLC system equipped with a UV detector
As a first step, the calibration curve is built by developing a validated HPLC method considering the following parameters: UV absorption characteristics of the analyte, mobile phase, column temperature, column pressure and injection volume.
The calibration curve has to consist of at least five different concentrations (in triplicate) ranging from a concentration higher than expected for the radiotracer down to the limit of quantification (LOQ). Plotting the AUCs (y-axis) versus compound concentration (µmol/mL, x-axis) and subsequent linear regression analysis requires a high R2-value in order to prove the linearity of the HPLC method. Our recommendation is a R2-value > 0.95. The injection of the formulation containing the radiotracer, allows the determination of AUCs from which compound concentrations (µmol/mL) are derived using the calibration curve. When the amount of radioactivity of an exact volume of formulation containing the radiotracer is determined in a dose calibrator (GBq/mL), this value has to be corrected for radiochemical purity. That means, only the collected radioactivity corresponding to the product should be included in the calculation.
During method validation, it is crucial to inject the crude mixture from the radiotracer synthesis to assure that (radio)impurities can be identified and baseline separated from the product to enable correct quantification.
Molar activity is not a constant value and changes over time. Therefore, the activity can be decay corrected to a defined reference time point e.g. end of synthesis (EOS) or end of bombardment (EOB). However, for patient or animal application and dose calculation it is a necessity to calculate the molar activity at the time point of administration.
Alternatively, if the mass limit is known, the maximum injectable mass can be specified based on volume i.e. “do not administer more than X mL”. Using this strategy is not necessary to calculate the Am at injection time. However, is it important that the volume of the end product is recorded accurately. Of course, consultation with the end users is crucial before embarking on a scanning campaign, so that all these issues can be agreed in advance of the scanning sessions.
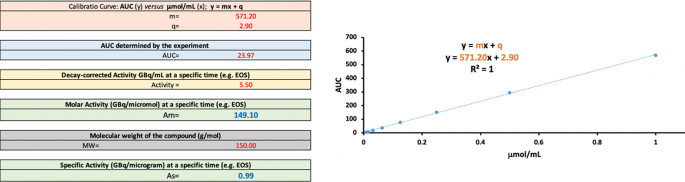
Example of how to calculate Am and As using Excel: Having a calibration curve of y = 571.20 x + 2.90 and a solution with an AUC of 23.97, the non-radioactive compound concentration present in the formulation is 0.04 µmol/mL. With an ActivityA of 5.50 GBq/mL at EOS, the Am at EOS is 149.1 GBq/µmol. The As (GBq/µg) is derived from Am by dividing by the molecular weight of the compound (Fig. 2, MW = 150, As = 0.99 GBq/µg at EOS).
Important in the context of Am calculation is the limit of quantification (LOQ) as evaluated during the method validation process. In radiochemistry it is common, that product concentrations below the LOQ are measured at the end of radiosynthesis. Here, the most correct value for product amount is to use the LOQ for the calculation instead of extrapolation of the data, followed by the statement that ‘Am is greater or equal to the calculated value’. An extrapolation beyond the linearity and limitation of an analytical method used for the calculation of the mass or molar concentration of the final product may overestimate the respective As and Am and must be avoided.
Examples of variability of molar activities for clinically applied PET radiotracers in different production laboratories
In general, molar activities play a crucial role for all PET radiotracers targeting saturable binding sites (e.g. receptors) but are secondary or negligible for many metabolic PET radiotracers where the endogenous levels of the compound are present in great excess of the radiotracer itself. The fluctuation in Am for carbon-11 labelled compounds is mainly caused by contamination with varying amounts of carrier carbon. A consistent small amount will come from the target gas but ingress of atmospheric carbon dioxide into the cyclotron target cannot be totally precluded and this will reduce the Am of the radiosynthon [11C]CO2 or [11C]CH4. Small amounts of carrier may also come from target body materials, valves, tubing etc. Similar challenges exist when processing radiometals and their contamination by non-radioactive metal contaminants. The examples below focus on carbon-11 PET radiotracers for comparison of Am derived from different laboratories.
Am varies between laboratories and radiotracers as exemplified in Fig. 3 for the PET radiotracers [11C]PIB (Philippe et al. 2011; Coliva et al. 2015; Buccino et al. 2019; Zhou et al. 2007; Verdurand et al. 2008; Boudjemeline et al. 2017), [11C]raclopride (Kawashima et al. 2015; Ishiwata et al. 1999, 2001; Oswald et al. 2005; Wilson et al. 2000; Andersson et al. 2009; Lee et al. 2016; Fujimura et al. 2010; Volkow et al. 2008; Alexoff et al. 2003; Schiffer et al. 2005; Farde et al. 1986; Mauger et al. 2005), [11C]choline (Biasiotto et al. 2012; Rosen et al. 1985; Cheung and Ho 2009; Reischl et al. 2004; Hara et al. 1997; Hara and Yuasa 1999) and [11C]verapamil (Elsinga et al. 1996; Hendrikse et al. 1998, 2001; Ikoma et al. 2006; Klerk et al. 2009; Assema et al. 2012; Wagner et al. 2011; Wegman et al. 2002; Luurtsema et al. 2003, 2005; Sasongko et al. 2005; Lubberink et al. 2007; Nagatani et al. 2001; Takano et al. 2006). Based on this literature, the common range for molar activities is 1 to 230 GBq/µmol (calculated at EOS) applying the ‘wet’ or gas phase method of [11C]methyl iodide production. The variation of Am are higher for [11C]PIB and [11C]choline than for [11C]verapamil. [11C]Raclopride, in this example, is an exception as two particularly high molar activities are found in literature. It is stated, that these higher molar activities can be achieved by applying a [11C]methane cyclotron target (Andersson et al. 2009; Fujimura et al. 2010; Noguchi and Suzuki 2003).
Examples highlighting the importance of high Am
Examples of highly toxic molecules: [11C]carfentanil, (+)-[18F]flubatine and (+)-[11C]PHNO
[11C]Carfentanil is a PET radiotracer used for imaging studies of µ-opioid receptors. The radiotracer is widely used to investigate the role of the of µ-opioid system in relation to obesity, pain regulation, drug addiction and many other disorders (Blecha et al. 2017). Carfentanil is approximately a thousand times more potent than morphine (Daele et al. 1976) Even at very low doses, unlabelled carfentanil can elicit a pharmacological response. Therefore, the mass limit is set to ≤ 0.03 µg/kg as an accepted standard (Blecha et al. 2017). This means that, for a clinical PET scan of an average subject of 70 kg, the calculated maximum administered mass is 2.1 µg (5.3 nmol). A high Am of [11C]carfentanil is therefore required to use this radiotracer safely. An injection dose of 400 MBq with 2.1 µg of carrier, can only be achieved if the Am of > 75 GBq/µmol at injection time (Perkins et al. 2014).
Many groups have developed radioligands targeting α4β2 as they are involved in various CNS disorders, Alzheimer’s and Parkinson’s neurodegenerative diseases, Tourette syndrome, anxiety, pain and depression. Development of PET radiotracers were mostly based on the epibatidine scaffold, which displays high toxicity (Mu et al. 2006). Structural modulations have been performed to improve affinity and selectivity in order to reduce toxicity. Nevertheless, these compounds still display toxic effects. For example, (+)-flubatine has a NOEL (no observed effect level) of 1.55 µg/kg in humans (Mu et al. 2006). The NOEL value requires the administration of (+)-[18F]flubatine with a Am exceeding 700 GBq/µmol in order to maintain a 1000-fold safety margin for the use of the radiotracer in clinical studies (Smits et al. 2014).
The potent dopamine D2/D3 receptor agonist (+)-PHNO ((+)-4-propyl-3,4,4a,5,6,10b-hexahydro-2H-naphtho[1,2-b][1,4]oxazin-9-ol, molecular weight 247.33 g/mol) can cause nausea, vomiting and orthostatic hypotension, at doses of 0.25 mg three times a day (Weiner et al. 1989). In initial clinical studies using the radiotracer (+)-[11C]PHNO, participants commonly experienced nausea (Willeit et al. 2006, 2008; Chiuccariello et al. 2013). Houle and co-workers subsequently recommended a dose below 0.029 µg/kg for (+)-[11C]PHNO, and therefore a maximum administered dose of 2.03 µg for a 70 kg participant, which is even lower than for [11C]carfentanil (Blecha et al. 2017). In this case, a molar activity of > 48.5 GBq/µmol at time of injection is required to avoid any side effects.
Example for reduced image quality based on apparent molar activity: [11C]mHED
Metaraminol, the precursor of the radiotracer [11C]meta-hydroxyephedrine ([11C]mHED (Mizrahi et al. 2009)) is authorised for the treatment of acute hypotension (Rosenspire et al. 1990). Both metaraminol as well as mHED itself can lead to increase of both systolic and diastolic blood pressure, therefore the sum of both compounds has to be evaluated after purification of the radiotracer and considered for safe administration of [11C]mHED (apparent Am) (Khavandi et al. 2009). Doses of 0.25–1 mg of metaraminol (Vraka et al. 2019) were safe to inject to patients. In most PET measurements, a pharmacological effect must be avoided and therefore a dose below a pharmacological or even toxic dose is a necessity (microdosing concept) (Critchley et al. 1999). In literature, the NOEL for mHED is not described, therefore it is recommended to include a safety margin for the calculation of the maximal applicable dose for a PET application. If calculated with e.g. a safety margin of 100 for the administration of [11C]mHED a maximal absolute dose of 2.5–10 µg (sum of mHED and metaraminol) per administration can be used as a limit to avoid any pharmacological effects during a PET-scan. Even at non-toxicological doses of mHED and metaraminol, it has been reported that doses of approximately 50 nmol/kg result in a decrease in myocardial uptake in mice (Bergström et al. 2003). This can be attributed to saturation of the norepinephrine receptors in the myocardium (competition between [11C]mHED and non-labelled mHED and metaraminol), and thus is a good example of the importance of maintaining high molar activities and apparent molar activities.
Examples highlighting where high Am is less important or detrimental
[18F]FDG and labelled endogenous molecules
Endogenous molecules, like amino acids, fatty acids, choline and glucose, labelled with radionuclides, undergo competition with the corresponding endogenous compounds in blood/tissue. Therefore, high Am is not usually required for these radiotracers. Although [18F]FDG is not an endogenous molecule, its uptake is related to the glucose levels in plasma. [18F]FDG with low Am does not result in deterioration of the PET image quality. However, high blood glucose levels do influence the radiotracer behaviour significantly, requiring strict adherence to fasting regimen for patients undergoing [18F]FDG scans (Law et al. 2010).
[111In]J591 → low Am is required
If Am plays a role in terms of pharmacological or even toxic effects, image quality has to be verified for each and every radiotracer. Especially for antibodies, it has been reported that the administration of [111In]J591 with low Am, achieved by adding unlabelled antibody, benefits the residence times and lesion to non-lesion residence time ratios (Trifiro and Paganelli 2007). The study suggests that higher administered antibody masses saturate the liver uptake and thus increase the biological half-life of the radiotracer. However, no standardised studies are yet available investigating the correlation of Am and image quality (signal-to-noise, standardized uptake value—SUV) for common radiotracers, requiring further research.
[18F]FDOPA → conflicting results- discussion about the role of Am
6-[18F]fluoro-3,4-dihydroxy-L-phenylalanine ([18F]FDOPA) has clinical indications in neurology and oncology. Kuik et al. (Pandit-Taskar et al. 2009) reported in a neuroendocrine tumour model study the use of [18F]FDOPA administered at doses spanning 3 orders of magnitude (11–33,000 GBq/µmol). In this study, the authors reported no significant difference in mean SUV and no statistically significant differences between the uptake values in biodistribution experiments between high and low Am groups. In conclusion, the authors claimed that even a 3000-fold difference in Am produced no significant differences in signal. Nevertheless, a similar clinical study using very high Am [18F]FDOPA (4000 GBq/µmol), showed higher uptake values compared to previous studies with significantly lower Am (Kuik et al. 2015). The authors indicated that the increased striatal-to-occipital-cortex ratio (SOR) observed could have been due to either the higher spatial resolution of the PET/CT scanner used, or the higher Am of the radiotracer in comparison to the previous studies. In addition to the potential effect on the image quality, low Am [18F]FDOPA can cause carcinoid crisis and flushes (Akamatsu et al. 2017 Feb) (Fig. 4).
Examples displaying the connection of molar activity, PET image quality and pharmaceutical dose. (left) [11C]mHED, requires high Am for sufficient quality of the PET scan. Before a pharmacological dose is reached the image quality is affected. (middle) [11C]PHNO requires high Am for patient application and safety. At first a pharmacological effect, like nausea, is observed before the quality of the PET image is influenced. The release criteria has to be strictly followed for this kind of compounds to avoid any side or toxic effects. (right) [111In]J591 is reported to have a low lesion to non-lesion residence time if the molar activity is high. Higher amounts of non-labelled autologue increases the residence time and subsequently the quality of the image
Influence of materials and beam parameters on Am and As
The selection and the use of appropriate materials and optimal beam performance are important to obtain high Am and As. Below is a list of critical items which may have an influence on Am and As.
-
a.
Quality of irradiated starting material. The production of most common radionuclides is performed using a cyclotron equipped with specially designed targets filled with starting materials. It is evident that staring materials must be highly pure to obtain high Am and As. Impurities and carriers can influence Am and As.
-
b.
Quality of gasses: Gas targets are used for the production of [11C]CO2 or [18F]F2 and filled with highly pure gas (99.999%) mixture of N2 + O2 (0.05–2%) or Ne or [18O]O2, respectively.
-
c.
Quality of liquids: For the production of [18F]NaF liquid targets are used, filled with [18O]H2O. Reusing of the target water can reduce the Am.
-
d.
Quality of generators: For generator-based radiometals the frequency of elution affects the apparent Am, as over the time the amount of the granddaughter decay product increases, e.g. 68Zn for 68 Ga/68Ge generators.
-
e.
Other parameters such as, irradiation time and beam current can also have influence on Am and As. For radiometals, these parameters are dependent on the producer of the radiometal or generator and cannot by influenced by the radiochemistry method per se.
-
f.
Target maintenance and cleaning. See for procedures in IAEA document https://www-pub.iaea.org/MTCD/Publications/PDF/Pub1563_web.pdf
Recommendations to obtain high Am and As
Several recommendations about target gas, columns to trap contaminations, target handling and general comments to support high Am and As. are listed in Table 1.
Pitfalls related to carbon-11 radiotracer production (what to do when introduction of carrier occurs?)
-
Check and analyse target gasses for carrier
-
Check leakage in the [11C]CH3I system. Air contains CO2 (380 ppm)
-
Check precursor and chemicals for carrier
-
Optimise synthesis times to increase radiochemical yield
-
Optimise preparative HPLC method to avoid traces of labelling precursors and other impurities in the final product.
Radiometals and Am
Over the last two decades, an increasing number of radiotracers have been labelled with radiometals for the personalized management of cancer patients. The radiotracers are usually labelled with an imaging radionuclide for the diagnosis, dosimetry and treatment planning, followed by labelling with a beta- or alpha-emitting radionuclide (e.g. 177Lu or 225Ac) for therapeutic applications. Such an approach corresponds to the so-called ‘theranostic’ paradigm.
In this section, we emphasized on gallium-68, however the following considerations can also be applied to other radiometals. Gallium-68 is increasingly used for labelling small biomolecules, such as peptides and small antibody fragments, and it is one of the main generator-based positron-emitting radionuclides currently used for PET imaging. 68 Ga is therefore particularly convenient because its production does not require an on-site cyclotron. Moreover, its 68 min half-life matches the rapid uptake kinetics of most radiolabelled peptides. Additionally, the ‘short’ half-life of gallium-68 and the rapid clearance of the radiolabelled peptides provide a suitably ‘low’ radiation dose to the patient. Consequently, several 68 Ga-labelled peptides have been developed to image various targets in tumour lesions, such as the somatostatin receptor subtype 2 ([68 Ga]Ga-DOTA-TATE), the prostate specific membrane antigen ([68 Ga]Ga-PSMA-617), the chemokine receptor type 4 ([68 Ga]Ga-Pentixafor), the gastrin releasing peptide receptor ([68 Ga]Ga-NeoBOMB1) and integrin αvβ3 ([68 Ga]Ga-NOTA-PRGD2) (Koopmans et al. 2005).
Gallium-68 complexation chemistry has been studied extensively and several bifunctional chelators are commercially available allowing stable 68 Ga-chelator complexes and conjugation to the peptide at the terminus position or on a specific functionality of amino acid side chain (i.e. lysine, cysteine) (Tanzey et al. 2018). Radiolabelling conditions have been optimized to warrant the robustness of the 68 Ga-labelling (Price and Orvig 2014). Indeed, the radiotracer preparation is generally fast and the incorporation of gallium-68 is nearly quantitative. Separation of the final radiotracer from the peptide precursor or the radionuclide is typically avoided during the radiochemical process. However, in the scenario of an incomplete radiometal incorporation, a simple purification of the radiotracer via solid phase extraction can be carried out to remove unreacted radionuclide. The main factor influencing the Am of a radiometallated peptide is the presence of peptide precursor and metal ion contaminants. The latter can compete with gallium-68 during the complexation reaction with the chelator. Thus, metal ion contaminants indirectly affect Am by necessitating higher amounts of peptide precursor to achieve high radionuclide incorporation. Rigorous elimination of metal impurities during the synthesis of the chelator-peptide conjugate, production and isolation of the radionuclide, and the radiolabelling is compulsory to obtain the highest achievable Am (Velikyan 2014). In fact, we are normally referring to the apparent Am for such radiotracers, empirically determined by dividing the amount of radioactivity obtained by the amount of peptide precursor engaged in the labelling process, since the final formulation of the labelled radiotracer contains remaining precursor and peptide complexed with metal impurities.
The requirement for high Am is however dictated by several biological factors, such as the affinity of the peptide for its receptor and the number of available receptors, limiting the mass dose of peptide that can be administered. Indeed, a suboptimal Am could result in excessive amount of peptide leading to the saturation of the receptor or eventually to the induction of pharmacological side effects, as it has been previously observed with GRPR agonists (Velikyan et al. 2008). Besides, the concomitant presence of unlabelled and labelled peptide binding to the same receptor can hamper a sufficient accumulation of radioactivity in the target tissue, and therefore deteriorate the quality of the PET images. Hence, a high Am is of paramount importance when developing PET radiotracers for saturable regulatory peptide binding processes or highly toxic molecules. Nevertheless, there are also situations, such as the somatostatin receptor-positive tumours, where the administration of an optimized dose of radiolabelled peptide is required to improve the sensitivity of detection. Indeed, it has been demonstrated that the uptake in somatostatin receptor-positive tumours is a bell-shaped function of the injected mass of peptide. With a higher administered mass dose of peptide, an increase of the tumour uptake is initially observed, most likely due to a faster rate of internalization by ligand-induced receptor clustering, followed by a drop in the tumour accumulation caused by the saturation of the receptor (Breeman et al. 2011).
In summary, there is no consensus on the Am for radiolabelled peptides, and an optimal value, that could be for instance a compromise between saturable elimination and receptor saturation, has to be experimentally defined.
Am considerations relating to a new generation of highly sensitive PET scanners: total body
Recent developments in PET scanner design have seen the emergence of a new generation of camera, the ‘total body’ PET scanners. By virtue of their extended detector coverage [up to two metres field of view cf. fifteen centimetres for a ‘typical’ PET-CT scanner these scanners currently report ca. five times greater sensitivity for dynamic imaging applications compared with ‘standard’ scanners (Breeman et al. 2001). The increase in sensitivity achievable with these tomographs may present new opportunities and challenges for the radiochemistry field.
The prospect of being able to administer lower doses of activity in total body PET scanning also implies that the administered radiotracer mass will be much lower. Currently, typical administered masses are often in the 2.5–25 nmol range. The injected mass of small molecule radiopharmaceuticals on classical PET–CT tomograph are normally in the range of 0.1–10 µg.
Assuming a fivefold increase in scanner sensitivity, administered radiotracer masses in the picogram range would be thus routinely achievable. This has several implications: (1) a significant decrease in administered radiotracer mass may enable the detection of poorly expressed molecular recognition sites [e.g. receptors, enzymes] not ‘imageable' using current practice as these molecular targets are significantly occupied by carrier tracer mass; (2) that radiotracers may be administered at a dose below the regulatory 1.5 µg ‘cut-off’ for unknown excipients in tablets and drug formulations (Cherry et al. 2018) requiring no traditional toxicology testing. This may be a way to enable translational studies at ‘sub-toxicological’ doses without the frequently encountered ‘toxicology testing cost barrier’ hampering first-in-human studies of otherwise promising novel radiotracers. If achievable, this could greatly enhance the productivity of new radiotracer evaluation and introduction for human use.
Footnote: Typically—for a small molecule radiotracer with a molecular weight 400, injected activity of 200 MBq and a molar activity of 10 GBq/µmol this corresponds to an injected mass of 8 µg (20 nmol).
For more information about toxicity of tracers, see position paper https://ejnmmipharmchem.springeropen.com/articles/10.1186/s41181-016-0004-6.
Conclusions
The Am or As of radiopharmaceuticals are important parameters and has an impact on safety and imaging quality This guideline will assist professionals to correctly define and measure molar activity (Am) and specific activity (As).
Availability of data and material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- Am :
-
Molar activity
- As :
-
Specific activity
- Bq:
-
Becquerel
- [18F]FDOPA:
-
6-[18F]fluoro-3,4-dihydroxy-L-phenylalanine
- GC/MS:
-
Gas chromatography/mass spectrometry
- HPLC:
-
High-performance liquid
- LC/MS:
-
Liquid chromatography/mass spectrometry
- NCA:
-
No-carrier added
- MW:
-
Molecular weight
- NOEL:
-
No observed effect level
- PET:
-
Positron emission tomography
- SOR:
-
Striatal-to-occipital-cortex ratio
- SUV:
-
Standard uptake value
- UPLC:
-
Ultra-performance liquid chromatography
References
Akamatsu G, Ohnishi A, Aita K, Nishida H, Ikari Y, Sasaki M, Kohara N, Senda M. A revisit to quantitative PET with 18F-FDOPA of high specific activity using a high-resolution condition in view of application to regenerative therapy. Ann Nucl Med. 2017;31(2):163–71.
Alexoff DL, Vaska P, Marsteller D, Gerasimov T, Li J, Logan J, et al. Reproducibility of 11C-raclopride binding in the rat brain measured with the microPET R4: effects of scatter correction and tracer specific activity. J Nucl Med. 2003;44(5):815–22.
Andersson J, Truong P, Halldin C. In-target produced [11C]methane: increased specific radioactivity. Appl Radiat Isot. 2009;67(1):106–10.
Balaram G, Munjal P, Dhagash AP. Validation of analytical procedures: methodology ICH-Q2B. Int J Pharm Innov. 2011;2249–1031(1):45–50.
Bergström M, Grahnén A, Långström B. Positron emission tomography microdosing: a new concept with application in tracer and early clinical drug development. Eur J Clin Pharmacol. 2003;59(5–6):357–66.
Biasiotto G, Bertagna F, Biasiotto U, Rodella C, Bosio G, Caimi L, et al. Description of high purity and high specific activity of [11C]Choline synthesis using TRACERlab FXc module, and detailed report of quality controls. Med Chem (los Angeles). 2012;8(6):1182–9.
Blecha JE, Henderson BD, Hockley BG, VanBrocklin HF, Zubieta JK, DaSilva AF, et al. An updated synthesis of [11C]carfentanil for positron emission tomography (PET) imaging of the μ-opioid receptor. J Label Compd Radiopharm. 2017;60(8):375–80.
Boudjemeline M, Hopewell R, Rochon PL, Jolly D, Hammami I, Villeneuve S, et al. Highly efficient solid phase supported radiosynthesis of [11C]PiB using tC18 cartridge as a “3-in-1” production entity. J Label Compd Radiopharm. 2017;60(14):632–8.
Breeman WA, De Jong M, Kwekkeboom DJ, Valkema R, Bakker WH, Kooij PP, et al. Somatostatin receptor-mediated imaging and therapy: basic science, current knowledge, limitations and future perspectives. Eur J Nucl Med. 2001;28(9):1421–9.
Breeman WAP, De Blois E, Sze Chan H, Konijnenberg M, Kwekkeboom DJ, Krenning EP. 68Ga-labeled DOTA-peptides and 68Ga-labeled radiopharmaceuticals for positron emission tomography: current status of research, clinical applications, and future perspectives. Semin Nucl Med. 2011;41(4):314–21.
Buccino P, Savio E, Porcal W. Fully-automated radiosynthesis of the amyloid tracer [11C] PiB via direct [11C]CO2 fixation-reduction. EJNMMI Radiopharm Chem. 2019;4(1):1–15.
Cherry SR, Jones T, Karp JS, Qi J, Moses WW, Badawi RD. Total-body PET: maximizing sensitivity to create new opportunities for clinical research and patient care. J Nucl Med. 2018;59(1):3–12.
Cheung MK, Ho CL. A simple, versatile, low-cost and remotely operated apparatus for [11C]acetate, [11C]choline, [11C]methionine and [11C]PIB synthesis. Appl Radiat Isot. 2009;67(4):581–9.
Chiuccariello L, Boileau I, Guranda M, Rusjan PM, Wilson AA, Zawertailo L, et al. Presentation of smoking-associated cues does not elicit dopamine release after one-hour smoking abstinence: A [11 C]-(+)-PHNO PET study. PLOS ONE. 2013;8(3):1–7.
Coenen HH, Gee AD, Adam M, Antoni G, Cutler CS, Fujibayashi Y, et al. Consensus nomenclature rules for radiopharmaceutical chemistry—setting the record straight. Nucl Med Biol. 2017;55:5–11.
Coliva A, Monterisi C, Apollaro A, Gatti D, Penso M, Gianolli L, et al. Synthesis optimization of 2-(4-N-[11C]methylaminophenyl)-6-hydroxybenzothiazole ([11C]PIB), amyloid PET imaging tracer for Alzheimer’s disease diagnosis. Appl Radiat Isot. 2015;105:66–71.
Critchley LAH, Karmakar MK, Cheng JHH, Critchley JAJH. A study to determine the optimum dose of metaraminol required to increase blood pressure by 25% during subarachnoid anaesthesia. Anaesth Intensive Care. 1999;27(2):170–4.
De Klerk OL, Willemsen ATM, Roosink M, Bartels AL, Harry Hendrikse N, Bosker FJ, et al. Locally increased P-glycoprotein function in major depression: a PET study with [11C]verapamil as a probe for P-glycoprotein function in the bloodbrain barrier. Int J Neuropsychopharmacol. 2009;12(7):895–904.
Elsinga PH, Franssen EJF, Hendrikse NH, Fluks L, Weemaes AMA, Van Der Graaf WTA, et al. Carbon-11labeled daunorubicin and verapamil for probing P-glycoprotein in tumors with PET. J Nucl Med. 1996;37(9):1571–5.
Farde L, Hall H, Ehrin E, Sedvall G. Quantitative analysis of D2 dopamie receptor binding in the living human brain by PET. Science. 1986;231(4735):258–61.
Fujimura Y, Ito H, Takahashi H, Yasuno F, Ikoma Y, Zhang MR, et al. Measurement of dopamine D2 receptors in living human brain using [11C]raclopride with ultra-high specific radioactivity. Nucl Med Biol. 2010;37(7):831–5.
Hara T, Yuasa M. Automated synthesis of [11C]choline, a positron-emitting tracer for tumor imaging. Appl Radiat Isot. 1999;50(3):531–3.
Hara T, Kosaka N, Shinoura N, Kondo T. PET imaging of brain tumor with [methyl-11C]choline. J Nucl Med. 1997;38(6):842–7.
Hendrikse NH, Schinkel AH, De Vries EGE, Fluks E, Van Der Graaf WTA, Willemsen ATM, et al. Complete in vivo reversal of P-glycoprotein pump function in the blood-brain barrier visualized with positron emission tomography. Br J Pharmacol. 1998;124(7):1413–8.
Hendrikse NH, De Vries EGE, Franssen EJF, Vaalburg W, Van Der Graaf WTA. In vivo measurement of [11C]verapamil kinetics in human tissues. Eur J Clin Pharmacol. 2001;56(11):827–9.
Ikoma Y, Takano A, Ito H, Kusuhara H, Sugiyama Y, Arakawa R, et al. Quantitative analysis of 11C-verapamil transfer at the human blood-brain barrier for evaluation of P-glycoprotein function. J Nucl Med. 2006;47(9):1531–7.
Ishiwata K, Ishii SI, Senda M. An alternative synthesis of [11C]raclopride for routine use. Ann Nucl Med. 1999;13(3):195–7.
Ishiwata K, Kobayashi T, Kawamura K, Matsuno K, Senda M. [11C]Raclopride binding was reduced in vivo by sigma1 receptor ligand SA4503 in the mouse brain, while [11C]SA4503 binding was not by raclopride. Nucl Med Biol. 2001;28(7):787–92.
Kawashima H, Kimura H, Nakaya Y, Tomatsu K, Arimitsu K, Nakanishi H, et al. Application of microreactor to the preparation of C-11-labeled compounds via O-[11C]methylation with [11C]CH3I: rapid synthesis of [11C]raclopride. Chem Pharm Bull. 2015;63(9):737–40.
Khavandi A, Gatward JJ, Whitaker J, Walker P. Myocardial infarction associated with the administration of intravenous ephedrine and metaraminol for spinal-induced hypotension. Anaesthesia. 2009;64(5):563–6.
Koopmans KP, Brouwers AH, De Hooge MN, Van Der Horst-Schrivers AN, Kema IP, Wolffenbuttel BH, et al. Carcinoid crisis after injection of 6–18F- fluorodihydroxyphenylalanine in a patient with metastatic carcinoid. J Nucl Med. 2005;46(7):1240–3.
Kuik WJ, Kema IP, Brouwers AH, Zijlma R, Neumann KD, Dierckx RAJO, et al. In vivo biodistribution of no-carrier-added 6–18F-fluoro-3, 4-dihydroxy-L-phenylalanine (18F-DOPA), produced by a new nucleophilic substitution approach, compared with carrier-added 18F-DOPA, prepared by conventional electrophilic substitution. J Nucl Med. 2015;56(1):106–12.
Law MP, Schäfers K, Kopka K, Wagner S, Schober O, Schäfers M. Molecular imaging of cardiac sympathetic innervation by 11C-mHED and PET: From man to mouse? J Nucl Med. 2010;51(8):1269–76.
Lee J, Cheong IK, Lee SY. Successful application of a neutral organic base, 1,8-bis(tetramethylguanidino)naphthalene (TMGN), for the radiosynthesis of [11C]raclopride. Appl Radiat Isot. 2016;118:382–8.
Lubberink M, Luurtsema G, Van Berckel BNM, Boellaard R, Toornvliet R, Windhorst AD, et al. Evaluation of tracer kinetic models for quantification of P-glycoprotein function using (R)-[11C]verapamil and PET. J Cereb Blood Flow Metab. 2007;27(2):424–33.
Luurtsema G, Molthoff CFM, Windhorst AD, Smit JW, Keizer H, Boellaard R, et al. (R)- and (S)-[11C]verapamil as PET-tracers for measuring P-glycoprotein function: in vitro and in vivo evaluation. Nucl Med Biol. 2003;30(7):747–51.
Luurtsema G, Molthoff CFM, Schuit RC, Windhorst AD, Lammertsma AA, Franssen EJF. Evaluation of (R)-[11C]verapamil as PET tracer of P-glycoprotein function in the blood-brain barrier: Kinetics and metabolism in the rat. Nucl Med Biol. 2005;32(1):87–93.
Mauger G, Saba W, Hantraye P, Dolle F, Coulon C, Bramoullé Y, et al. Multiinjection approach for D2 receptor binding quantification in living rats using [11C]raclopride and the β-microprobe: Crossvalidation with in vitro binding data. J Cereb Blood Flow Metab. 2005;25(11):1517–27.
Mizrahi R, Wilson A, Houle S. Side effects profile of [11C]-(+)-PHNO in human, a dopamine D2/3 agonist ligand. J Nucl Med. 2009;50(2):1288.
Mu L, Drandarov K, Bisson WH, Schibig A, Wirz C, Schubiger PA, et al. Synthesis and binding studies of epibatidine analogues as ligands for the nicotinic acetylcholine receptors. Eur J Med Chem. 2006;41(5):640–50.
Nagatani G, Nomoto M, Takano H. Transcriptional activation of the human HMG1 gene in cisplatin-resistant human. Cancer Cells. 2001;1592–7.
Noguchi J, Suzuki K. Automated synthesis of the ultra high specific activity of [11C]Ro15-4513 and its application in an extremely low concentration region to an ARG study. Nucl Med Biol. 2003;30(3):335–43.
Oswald LM, Wong DF, McCaul M, Zhou Y, Kuwabara H, Choi L, et al. Relationships among ventral striatal dopamine release, cortisol secretion, and subjective responses to amphetamine. Neuropsychopharmacology. 2005;30(4):821–32.
Pandit-Taskar N, Donoghue JAO, Morris MJ, Wills EA, Schwartz LH, Gonen M, et al. Antibody mass escalation study in patients with castration resistant prostate cancer using 111I–J591: Lesion detectability and dosimetric projections for 90Y radioimmunotherapy. J Nucl Med. 2009;49(7):1066–74.
Perkins G, Sheth R, Greguric I, Pascali G. Optimisation of [11C]Raclopride production using a Synthra GPextent system. Curr Radiopharm. 2014;7(2):100–6.
Philippe C, Haeusler D, Mitterhauser M, Ungersboeck J, Viernstein H, Dudczak R, et al. Optimization of the radiosynthesis of the Alzheimer tracer 2-(4-N-[11C]methylaminophenyl)-6-hydroxybenzothiazole ([11C]PIB). Appl Radiat Isot. 2011;69(9):1212–7.
Price EW, Orvig C. Matching chelators to radiometals for radiopharmaceuticals. Chem Soc Rev. 2014;43(1):260–90.
Reischl G, Bieg C, Schmiedl O, Solbach C, Machulla HJ. Highly efficient automated synthesis of [11C]choline for multi dose utilization. Appl Radiat Isot. 2004;60(6):835–8.
Rosen MA, Jones RM, Yano Y, Budinger TF. Carbon-11 choline: synthesis. Uptake Inhibition by 2-Dimethylaminoethanol A 1985;26:1424–8.
Rosenspire KC, Haka MS, Van Dort ME, Jewett DM, Gildersleeve DL, Schwaiger M, et al. Synthesis and preliminary evaluation of carbon-11-meta-hydroxyephedrine: a false transmitter agent for heart neuronal imaging. J Nucl Med. 1990;31(8):1328–34.
Sasongko L, Link JM, Muzi M, Mankoff DA, Yang X, Collier AC, et al. Imaging P-glycoprotein transport activity at the human blood-brain barrier with positron emission tomography. Clin Pharmacol Ther. 2005;77(6):503–14.
Schiffer WK, Alexoff DL, Shea C, Logan J, Dewey SL. Development of a simultaneous PET/microdialysis method to identify the optimal dose of 11C-raclopride for small animal imaging. J Neurosci Methods. 2005;144(1):25–34.
Smits R, Fischer S, Hiller A, Deuther-Conrad W, Wenzel B, Patt M, et al. Synthesis and biological evaluation of both enantiomers of [18F]flubatine, promising radiotracers with fast kinetics for the imaging of α4β2-nicotinic acetylcholine receptors. Bioorganic Med Chem. 2014;22(2):804–12.
Takano A, Kusuhara H, Suhara T, Ieiri I, Morimoto T, Lee YJ, et al. Evaluation of in vivo P-glycoprotein function at the blood-brain barrier among MDR1 gene polymorphisms by using 11C-verapamil. J Nucl Med. 2006;47(9):1427–33.
Tanzey S, Thompson S, Scott P, Brooks AF. Gallium-68: methodology and novel radiotracers for positron emission tomography (2012–2017). Pharm Pat Anal. 2018;5:193–227.
Trifiro G, Paganelli G. [18F]FDG uptake: pay attention to candies. Ecancermedicalscience. 2007;1(48):2–5.
van Assema DME, Lubberink M, Boellaard R, Schuit RC, Windhorst AD, Scheltens P, et al. Reproducibility of quantitative (R)-[11C]verapamil studies. EJNMMI Res. 2012;2(1):1–10.
Van Daele PGH, De Bruyn MFL, Boey JM, Sanczuk S, Agten JTM, Janssen PAJ. Synthetic analgesics: N-(1-[2-arylethyl]-4-substituted 4-piperidinyl) N-arylalkanamides. Arzneimittelforschung. 1976;26(8):1521–31.
Velikyan I. Prospective of 68Ga-radiopharmaceutical development. Theranostics. 2014;4(1):47–80.
Velikyan I, Beyer GJ, Bergström-Pettermann E, Johansen P, Bergström M, Långström B. The importance of high specific radioactivity in the performance of 68Ga-labeled peptide. Nucl Med Biol. 2008;35(5):529–36.
Verbruggen A, Coenen HH, Deverre JR, Guilloteau D, Langstrom B, Salvadori PA, Halldin C. Guideline to regulations for radiopharmaceuticals in early phase clinical trials in the EU. Eur J Nucl Med Mol Imaging. 2008;35(11):2144–51.
Verdurand M, Bort G, Tadino V, Bonnefoi F, Le Bars D, Zimmer L. Automated radiosynthesis of the Pittsburg compound-B using a commercial synthesizer. Nucl Med Commun. 2008;29(10):920–6.
Volkow ND, Wang GJ, Telang F, Fowler JS, Logan J, Wong C, et al. Sleep deprivation decreases binding of [11C]raclopride to dopamine D2/D3 receptors in the human brain. J Neurosci. 2008;28(34):8454–61.
Vraka C, Pichler V, Berroterán-Infante N, Wollenweber T, Pillinger A, Hohensinner M, et al. Optimization of the automated synthesis of [11C]MHED—administered and apparent molar activities. Pharmaceuticals. 2019;12(1):12. https://doi.org/10.3390/ph12010012.
Wagner CC, Simpson M, Zeitlinger M, Bauer M, Karch R, Abrahim A, et al. A combined accelerator mass spectrometry-positron emission tomography human microdose study with 14C- and 11C-labelled verapamil. Clin Pharmacokinet. 2011;50(2):111–20.
Wegman TD, Maas B, Elsinga PH, Vaalburg W. An improved method for the preparation of [11C]verapamil. Appl Radiat Isot. 2002;57(4):505–7.
Weiner W, Factor S, Sanchez-Ramos J. The efficacy of (+)-4-propyl-9- hydroxynaphthoxazine as adjunctive therapy in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 1989;52:732–5.
Willeit M, Ginovart N, Kapur S, Houle S, Hussey D, Seeman P, et al. High-affinity states of human brain dopamine D2/3 receptors imaged by the agonist [11C]-(+)-PHNO. Biol Psychiatry. 2006;59(5):389–94.
Willeit M, Ginovart N, Graff A, Rusjan P, Vitcu I, Houle S, et al. First human evidence of d-amphetamine induced displacement of a D 2/3 agonist radioligand: A [11C]-(+)-PHNO positron emission tomography study. Neuropsychopharmacology. 2008;33(2):279–89.
Wilson AA, Garcia A, Jin L, Houle S. Radiotracer synthesis from [11C]-iodomethane: a remarkably simple captive solvent method. Nucl Med Biol. 2000;27(6):529–32.
Zhou Y, Resnick SM, Ye W, Fan H, Holt DP, Klunk WE, et al. Using a reference tissue model with spatial constraint to quantify [11C]Pittsburgh compound B PET for early diagnosis of Alzheimer’s disease. Neuroimage. 2007;36(2):298–312.
Acknowledgements
We thank the EANM committees, EANM national delegates for their review, suggestions and comments. The authors (G.L. and P.E.) thank Prof. dr. A.D. Windhorst for sharing his expertise on Am.
Preamble: The European Association of Nuclear Medicine (EANM) is a professional non-profit medical association that facilitates communication worldwide among individuals pursuing clinical and research excellence in nuclear medicine. The EANM was founded in 1985. This EANM guideline has been written by members who are selected on the basis of their scientific good standing and active engagement with relevant scientific societies. This guideline is intended to assist professionals to correctly define and measure molar activity (Am) and specific activity (As), discuss common pitfalls and good practice in the production and measurement of radiotracers with high Am and is written in accordance with the recently published consensus nomenclature rules (Coenen et al. 2017).
Scope: Several classes of compounds such as small molecules, peptides and macromolecules radiolabelled for diagnostic and therapeutic purposes.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
All authors contributed equally in the discussion and preparation of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luurtsema, G., Pichler, V., Bongarzone, S. et al. EANM guideline for harmonisation on molar activity or specific activity of radiopharmaceuticals: impact on safety and imaging quality. EJNMMI radiopharm. chem. 6, 34 (2021). https://doi.org/10.1186/s41181-021-00149-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41181-021-00149-6